News
Indiana Vein Specialists® Adds a New Treatment Method

Indiana Vein Specialists® announced recently that they are the first medical practice in the greater Indianapolis area to treat varicose vein patients with Varithena®, a newly released foam sclerosant medication that can be used to treat many forms of varicose vein disease. Varithena® has been under development for the last 14 years, demonstrating safety and efficacy in clinical trials. The drug was approved by the FDA in November 2013.
“Our clinical experience and the high quality of our ultrasound mapping are the primary reasons we are able to offer this treatment to selected patients as an alternative to other accepted methods of varicose vein treatment,” Indiana Vein Specialists®’ owner and chief medical officer Jeffery P. Schoonover, M.D.™, said. “Endovenous chemical ablation (ECA) with Varithena® allows for an additional minimally invasive treatment strategy in the management of chronic venous disease.”
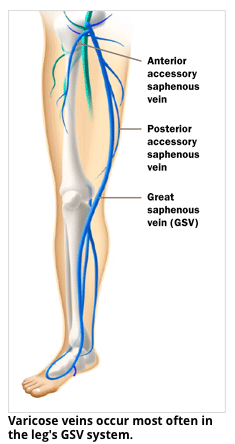
 Varithena® is approved by the FDA for treatment of the great saphenous vein and the accessory great saphenous vein, which are the largest superficial veins in the leg and usually most problematic. Varithena® is also indicated for visible varicose veins over three milimeters in diameter that feed into these saphenous veins. Treatment of the great saphenous vein or accessory saphenous vein with Varithena® involves injection of local anesthetic into the skin and then placement of a tiny needle and plastic catheter into the vein with ultrasound guidance. Varithena® is then injected into the vein with ultrasound guidance to direct the medication into the veins to be treated. The medication displaces the blood within the treated veins and causes the veins to seal shut. The abnormal veins then shrink and are absorbed by the body over the next several months. Patients resume walking immediately after the procedure and may resume most normal activities the day of the procedure. Treatment of varicose vein disease with Varithena® may require additional treatment sessions, depending on the initial presentation and severity of the varicose vein disease.
Varithena® is approved by the FDA for treatment of the great saphenous vein and the accessory great saphenous vein, which are the largest superficial veins in the leg and usually most problematic. Varithena® is also indicated for visible varicose veins over three milimeters in diameter that feed into these saphenous veins. Treatment of the great saphenous vein or accessory saphenous vein with Varithena® involves injection of local anesthetic into the skin and then placement of a tiny needle and plastic catheter into the vein with ultrasound guidance. Varithena® is then injected into the vein with ultrasound guidance to direct the medication into the veins to be treated. The medication displaces the blood within the treated veins and causes the veins to seal shut. The abnormal veins then shrink and are absorbed by the body over the next several months. Patients resume walking immediately after the procedure and may resume most normal activities the day of the procedure. Treatment of varicose vein disease with Varithena® may require additional treatment sessions, depending on the initial presentation and severity of the varicose vein disease.
“Evaluation and treatment of venous disease is a complex process and every patient is unique,” Schoonover said. “Varithena® is not for every patient, but those who are troubled by varicose veins, soft tissue pain, leg aching or tenderness, or recurrent rashes and skin changes near the ankles should seek a consultation. We will evaluate your problem, educate you regarding lifestyle changes and provide you with a detailed review of your treatment options.”
Those who wish to learn more about vein problems are encouraged to visit the website at www.indyveins.com or to call 317-348-3020 for a consultation appointment.
Indiana Vein Specialists® is a medical practice which provides comprehensive, office-based and minimally invasive care of venous insufficiency. Venous disease is a chronic condition and IVS believes a continuity based care model is the optimal way to approach the spectrum of venous insufficiency. The practice’s mission is twofold: the venous health of your legs today and the venous health of your legs tomorrow, utilizing the best available tools, training, and technology to accomplish this goal. Indiana Vein Specialists® has two locations to serve patients: 118th and Olio Road in Fishers and 106th and Michigan Road in the West Carmel/Zionsville area.
